12 Achilles Tendon and Heel Appearance Changes Linked to Metabolic Disorders
# 12 Achilles Tendon and Heel Appearance Changes Linked to Metabolic Disorders: A Comprehensive Medical Analysis
The human foot and ankle complex serves as a remarkable window into systemic health, with the Achilles tendon and heel region functioning as sensitive indicators of underlying metabolic dysfunction. Recent advances in medical research have illuminated a profound connection between various metabolic disorders and distinctive changes in the appearance, structure, and function of these critical lower extremity components. From the crystalline deposits of gout that create characteristic nodular deformities to the glycation-induced stiffening seen in diabetes mellitus, metabolic disturbances manifest in predictable and diagnostically significant ways throughout the heel and Achilles tendon complex. This comprehensive analysis explores twelve specific appearance changes that serve as clinical markers for metabolic disorders, ranging from the subtle early signs of insulin resistance to the dramatic structural alterations associated with advanced endocrine dysfunction. Understanding these connections empowers healthcare providers to recognize systemic disease through careful examination of the lower extremities, while simultaneously helping patients appreciate how their metabolic health directly impacts their mobility and quality of life. The following detailed exploration reveals how conditions such as diabetes, thyroid disorders, lipid abnormalities, and inflammatory metabolic syndromes leave their distinctive signatures on the heel and Achilles tendon, creating a roadmap for early detection, intervention, and comprehensive metabolic management.
1. Diabetic Achilles Tendon Thickening and Glycation-Induced Changes
Diabetes mellitus produces some of the most recognizable and clinically significant changes in Achilles tendon morphology, primarily through the process of advanced glycation end product (AGE) formation. In diabetic patients, chronically elevated blood glucose levels lead to non-enzymatic glycation of collagen fibers within the tendon structure, resulting in cross-linking that fundamentally alters the mechanical properties and appearance of the tissue. This biochemical transformation manifests as progressive tendon thickening, with diabetic patients showing Achilles tendons that are typically 20-30% thicker than those of non-diabetic individuals. The affected tendons develop a characteristic rope-like appearance with loss of the normal subtle undulations visible on ultrasound imaging. Additionally, the tendon's echogenicity changes, appearing more heterogeneous and hyperechoic due to the accumulated glycation products and associated inflammatory responses. Clinically, patients may notice decreased ankle flexibility, morning stiffness that persists longer than typical age-related changes, and a gradual loss of the springy quality that normally characterizes healthy tendon function. The glycation process also compromises the tendon's ability to repair micro-injuries, leading to a higher susceptibility to rupture and slower healing times when injuries do occur. These changes often precede the development of more obvious diabetic complications, making Achilles tendon assessment a valuable tool for monitoring glycemic control and predicting future diabetic complications.
2. Gouty Tophi Formation and Uric Acid Crystal Deposition
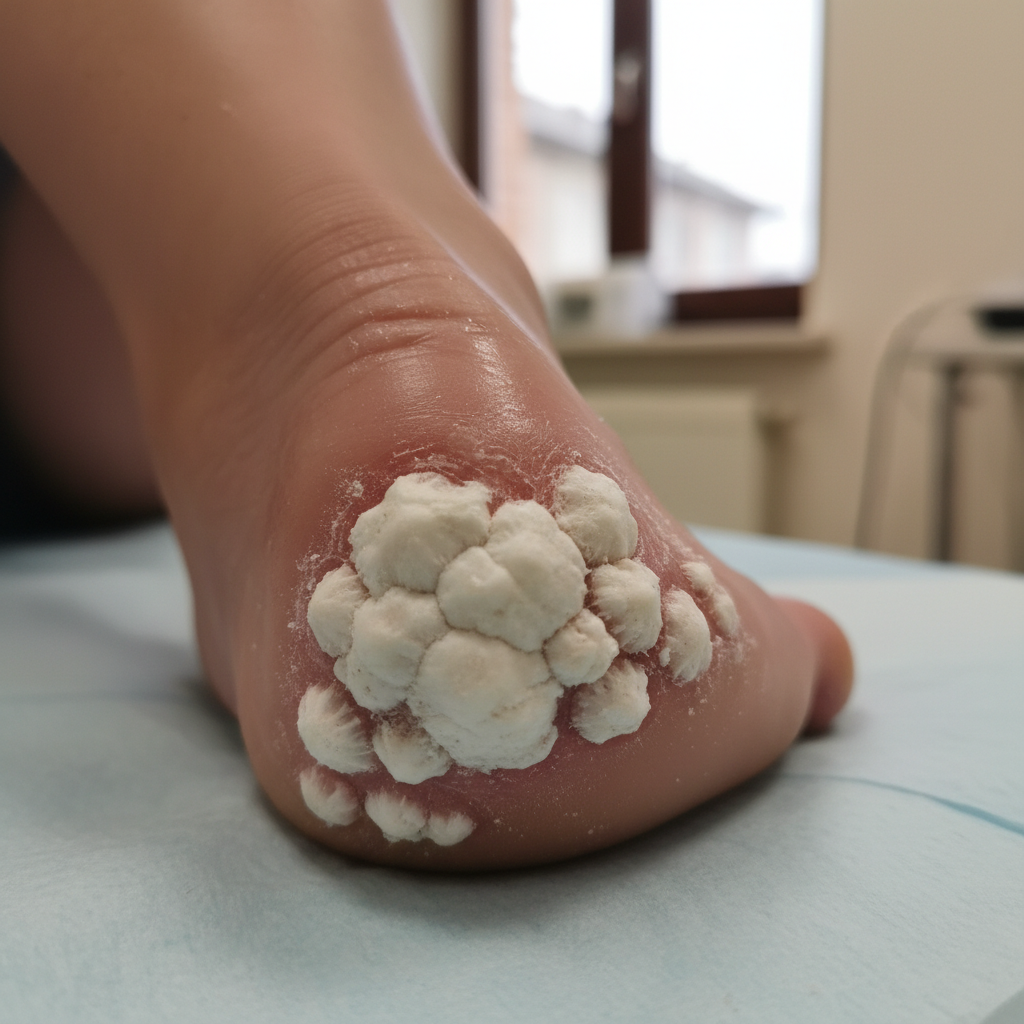
Gout, a metabolic disorder characterized by elevated serum uric acid levels, creates some of the most visually distinctive changes in the heel and Achilles tendon region through the formation of tophi—nodular deposits of monosodium urate crystals. These crystalline accumulations typically appear as firm, whitish or yellowish nodules that can range from small, barely palpable bumps to large, deforming masses that significantly alter the normal contour of the Achilles tendon and surrounding tissues. The tophi often develop along the tendon's insertion point at the calcaneus, creating a characteristic "lumpy" appearance that may be accompanied by overlying skin changes including thinning, discoloration, and occasional ulceration when the deposits approach the surface. Advanced cases may show chalk-like material extruding through the skin, a pathognomonic sign of chronic tophaceous gout. The formation of these deposits follows a predictable pattern, typically beginning with microscopic crystal accumulation that gradually coalesces into visible nodules over months to years of sustained hyperuricemia. Patients frequently report that these areas become acutely painful and inflamed during gout flares, with the surrounding tissue becoming red, swollen, and exquisitely tender. The mechanical effects of tophi can significantly impair Achilles tendon function, leading to decreased plantarflexion strength and altered gait mechanics. Importantly, the presence of tophi indicates chronic, poorly controlled gout and suggests an increased risk for joint destruction and kidney complications, making their recognition crucial for initiating appropriate urate-lowering therapy.
3. Thyroid-Related Tendon Swelling and Myxedematous Changes
Thyroid disorders, particularly hypothyroidism, produce characteristic changes in the Achilles tendon and heel region that reflect the systemic effects of altered thyroid hormone metabolism. In hypothyroid patients, decreased thyroid hormone levels lead to the accumulation of mucopolysaccharides, particularly hyaluronic acid, within connective tissues—a condition known as myxedema. This biochemical alteration manifests in the Achilles tendon as diffuse, non-pitting swelling that gives the tendon a thickened, doughy consistency quite different from the firm, rope-like thickening seen in diabetes. The affected tendon loses its normal sharp definition and may appear to blend into the surrounding tissues, creating a characteristic "puffy" appearance that extends into the heel pad and surrounding soft tissues. Patients with severe hypothyroidism may develop what clinicians term "myxedematous heel," where the entire heel region becomes boggy and enlarged, often accompanied by a characteristic orange-peel texture of the overlying skin. The delayed relaxation phase of the Achilles reflex, a classic neurological finding in hypothyroidism, correlates with these structural changes and reflects the overall slowing of metabolic processes. Conversely, hyperthyroid patients may experience Achilles tendon inflammation and pain due to increased metabolic activity and autoimmune processes, though visible structural changes are less common. The reversible nature of these thyroid-related changes makes them particularly important to recognize, as appropriate thyroid hormone replacement therapy can restore normal tendon appearance and function, highlighting the critical relationship between endocrine balance and musculoskeletal health.
4. Xanthomatous Deposits in Familial Hypercholesterolemia
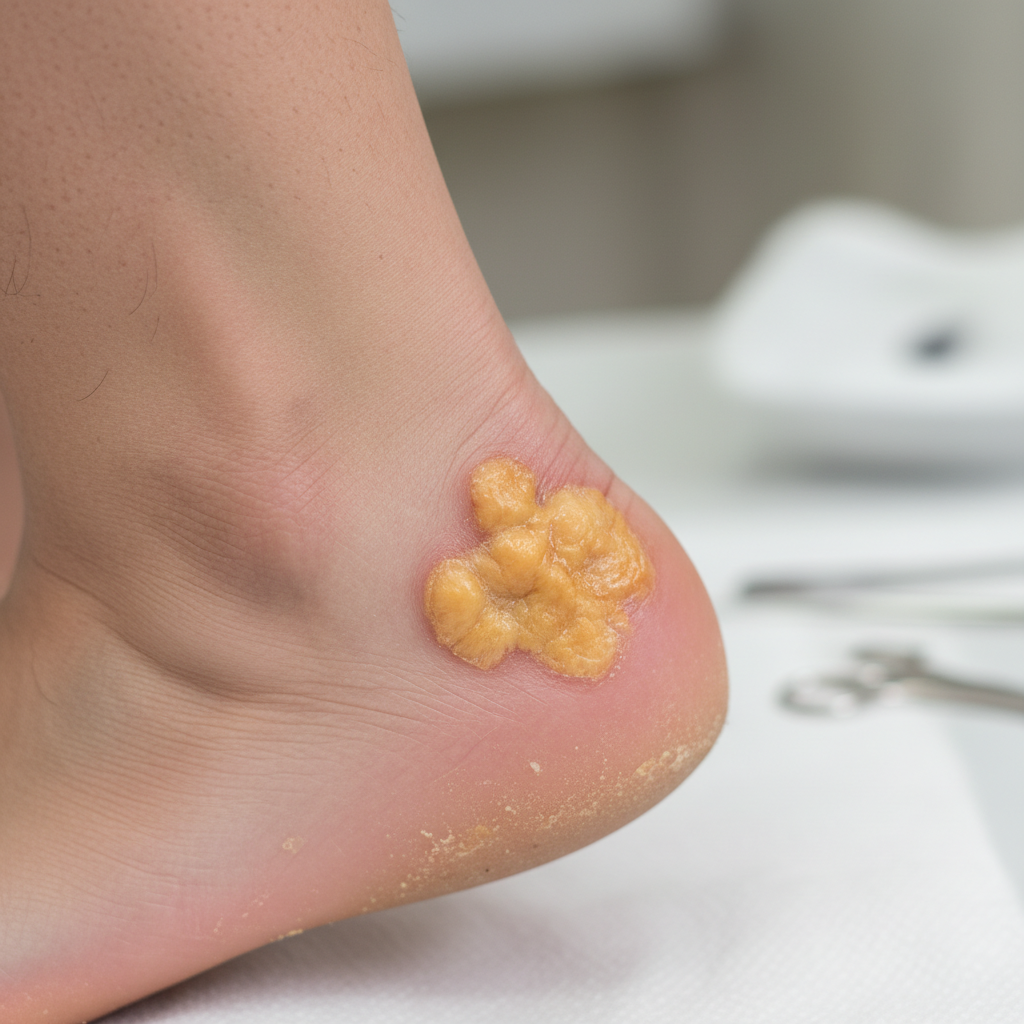
Familial hypercholesterolemia and other severe lipid disorders create distinctive yellow-orange deposits called xanthomas in the Achilles tendon and heel region, representing one of the most visually striking manifestations of metabolic dysfunction. These cholesterol-rich deposits typically appear as firm, yellowish nodules or plaques that may be confused with other tendon abnormalities but have characteristic features that distinguish them from conditions like gout or inflammatory tendinopathies. Achilles tendon xanthomas usually develop bilaterally and symmetrically, beginning as small, barely visible deposits that gradually enlarge over years of sustained hypercholesterolemia. The deposits often create a characteristic "lumpy" or "knotted" appearance along the tendon's length, with some patients developing massive xanthomatous involvement that significantly enlarges the tendon's cross-sectional area. The overlying skin may appear normal initially but can develop a yellowish discoloration as the deposits approach the surface, and in advanced cases, the xanthomas may ulcerate or become infected. These deposits are not merely cosmetic concerns but represent a significant cardiovascular risk marker, as patients with tendon xanthomas have markedly elevated rates of premature coronary artery disease and other atherosclerotic complications. The mechanical effects of large xanthomatous deposits can impair Achilles tendon function, leading to decreased ankle mobility and altered biomechanics that may predispose to injury. Importantly, aggressive lipid-lowering therapy can lead to regression of these deposits over time, making their recognition crucial for initiating appropriate cardiovascular risk reduction strategies and monitoring treatment effectiveness.
5. Calcaneal Spurs and Insulin Resistance Syndrome
The development of calcaneal spurs—bony projections from the heel bone—shows a strong association with insulin resistance and metabolic syndrome, representing a mechanical manifestation of systemic metabolic dysfunction. Research has demonstrated that patients with insulin resistance have significantly higher rates of both plantar and posterior calcaneal spur formation, likely due to the complex interplay between altered glucose metabolism, chronic inflammation, and biomechanical stress. Insulin resistance promotes a pro-inflammatory state characterized by elevated levels of cytokines such as TNF-alpha and IL-6, which can stimulate osteoblast activity and promote abnormal bone formation at sites of mechanical stress. The posterior calcaneal spur, which develops at the Achilles tendon insertion, creates a characteristic radiographic appearance of a hook-like bony projection that may be visible on physical examination as a palpable prominence at the back of the heel. These spurs often develop in conjunction with Achilles tendon thickening and may contribute to the development of insertional tendinopathy, creating a cycle of inflammation and mechanical dysfunction. Patients frequently report heel pain that is worse with initial weight-bearing after periods of rest, a symptom pattern that correlates with the degree of metabolic dysfunction. The plantar calcaneal spur, while not directly affecting the Achilles tendon, often coexists with posterior spurs and contributes to overall heel dysfunction. The recognition of calcaneal spurs in the context of metabolic assessment can serve as an early indicator of insulin resistance, prompting evaluation for diabetes risk factors and implementation of lifestyle interventions that may prevent progression to overt diabetes mellitus.
6. Achilles Tendon Rupture Risk in Diabetic Patients

Diabetes mellitus significantly increases the risk of Achilles tendon rupture through multiple pathophysiological mechanisms that compromise tendon integrity and healing capacity. The advanced glycation end products (AGEs) that accumulate in diabetic patients create abnormal cross-links between collagen fibers, making the tendon more brittle and less able to withstand normal mechanical stresses. This biochemical alteration, combined with diabetic microangiopathy that reduces blood flow to the tendon, creates a perfect storm for catastrophic tendon failure. Diabetic patients show rupture rates that are 2-4 times higher than non-diabetic individuals, with ruptures often occurring during relatively minor activities that would not typically cause injury in healthy individuals. The appearance of the Achilles tendon in diabetic patients at risk for rupture often shows characteristic warning signs including focal areas of thinning, loss of the normal fibrillar pattern on ultrasound, and the development of intratendinous calcifications that appear as bright, echogenic foci within the tendon substance. Pre-rupture changes may include the development of fusiform swelling in the mid-portion of the tendon, areas of decreased echogenicity indicating tissue degeneration, and loss of the normal parallel fiber arrangement. When rupture does occur in diabetic patients, the healing process is significantly impaired due to compromised angiogenesis, delayed cellular proliferation, and altered collagen synthesis. The clinical appearance of a diabetic Achilles rupture may be more subtle than in non-diabetic patients, with less obvious swelling and bruising due to impaired inflammatory responses, making diagnosis more challenging and potentially delaying appropriate treatment.
7. Peripheral Neuropathy and Heel Pad Changes

Diabetic peripheral neuropathy creates a cascade of changes in the heel pad and surrounding tissues that significantly alter the normal appearance and function of this critical weight-bearing structure. The loss of protective sensation that characterizes diabetic neuropathy leads to altered gait mechanics and abnormal pressure distribution during walking, resulting in characteristic changes to the heel pad's thickness, composition, and structural integrity. Neuropathic patients often develop heel pad atrophy, where the normal thick, fatty cushion that protects the calcaneus becomes thin and fibrotic, creating a characteristic "bony" prominence that is easily palpable through the skin. This atrophy occurs due to a combination of factors including decreased weight-bearing stimulus from altered gait patterns, impaired microcirculation, and the direct effects of chronic hyperglycemia on adipose tissue metabolism. The visual appearance of the neuropathic heel often shows skin changes including dryness, scaling, and the development of hyperkeratotic areas that reflect the loss of normal autonomic innervation to sweat glands and blood vessels. Patients may develop characteristic pressure ulcers over bony prominences, particularly over the posterior and plantar aspects of the calcaneus, which appear as circular, punched-out lesions with minimal surrounding inflammation due to the impaired pain response. The combination of heel pad atrophy and sensory loss creates a particularly dangerous situation where repetitive trauma can occur without the patient's awareness, leading to the development of chronic wounds that heal poorly due to the underlying metabolic dysfunction. Recognition of these early neuropathic changes in heel pad appearance can prompt intervention strategies including protective footwear, pressure redistribution devices, and aggressive glycemic control to prevent progression to more serious complications.
8. Lipodystrophy and Heel Fat Pad Abnormalities

Lipodystrophy syndromes, characterized by abnormal fat distribution and metabolism, create distinctive changes in the heel fat pad that can significantly alter both the appearance and function of the heel region. These rare metabolic disorders, which can be congenital or acquired, result in selective loss of subcutaneous fat from specific body regions, often including the heel pad, while causing abnormal fat accumulation in other areas. The heel fat pad in lipodystrophic patients typically appears markedly atrophic, with the normal thick, cushioning layer of specialized fat reduced to a thin, fibrotic structure that provides minimal protection to the underlying calcaneus. This creates a characteristic "bony heel" appearance where the calcaneal tuberosity is easily palpable and may be visible as a prominent contour beneath the skin. The overlying skin often appears tight and shiny due to the loss of underlying fat volume, and patients frequently develop hyperkeratotic areas over pressure points as the body attempts to compensate for the lost cushioning. The functional consequences of heel fat pad atrophy in lipodystrophy are significant, with patients experiencing pain during weight-bearing activities and an increased risk of developing pressure ulcers over bony prominences. Associated metabolic abnormalities in lipodystrophy, including severe insulin resistance and hypertriglyceridemia, may compound the local effects on heel appearance by promoting inflammation and impaired wound healing. The recognition of heel fat pad changes in the context of lipodystrophy is crucial for comprehensive management, as these patients require specialized care including pressure-redistributing footwear, metabolic monitoring, and treatment of associated complications such as diabetes and cardiovascular disease.
9. Inflammatory Arthropathy and Achilles Enthesitis
Inflammatory arthropathies, particularly those associated with metabolic dysfunction such as psoriatic arthritis and reactive arthritis, commonly affect the Achilles tendon insertion site, creating characteristic changes known as enthesitis. This inflammatory process at the tendon-bone interface produces distinctive clinical and imaging findings that reflect the underlying autoimmune and metabolic disturbances. The affected Achilles tendon insertion typically shows fusiform swelling that extends both proximally along the tendon and distally into the calcaneal region, creating a characteristic "sausage-like" appearance that distinguishes it from other causes of heel pain. The overlying skin may show erythema, warmth, and occasionally the development of psoriatic plaques in patients with psoriatic arthritis, providing important clues to the underlying systemic condition. Chronic enthesitis leads to characteristic radiographic changes including erosions at the tendon insertion site, new bone formation, and the development of enthesophytes—bony spurs that form as part of the inflammatory healing process. The inflammatory process often extends beyond the immediate insertion site to involve the retrocalcaneal bursa, creating a complex pattern of swelling and tenderness that can be distinguished from simple mechanical tendinopathy by its association with systemic inflammatory markers and response to anti-inflammatory treatments. Patients with metabolic syndrome and inflammatory arthropathy show particularly severe enthesitis, likely due to the synergistic effects of chronic inflammation and metabolic dysfunction on tendon healing and remodeling. The recognition of Achilles enthesitis as part of a systemic inflammatory condition is crucial for appropriate treatment, as these patients require immunosuppressive therapy rather than simple mechanical interventions, and the presence of enthesitis may indicate increased cardiovascular risk due to systemic inflammation.
10. Amyloidosis and Tendon Infiltration
Amyloidosis, a group of disorders characterized by the abnormal deposition of misfolded proteins, can significantly affect the Achilles tendon and heel region, creating distinctive changes that reflect the underlying metabolic dysfunction. Primary amyloidosis (AL type) and secondary amyloidosis associated with chronic inflammatory conditions both show predilection for tendon involvement, with the Achilles tendon being one of the most commonly affected sites. The deposition of amyloid fibrils within the tendon structure creates characteristic changes in both the mechanical properties and appearance of the tissue, with affected tendons showing progressive thickening and loss of normal elasticity. The clinical appearance of amyloid-infiltrated Achilles tendons is distinctive, with patients developing firm, non-tender swelling that has a characteristic "woody" consistency quite different from the soft, fluctuant swelling seen in inflammatory conditions or the rope-like thickening of diabetic tendons. The tendon may appear enlarged along its entire length, and in advanced cases, the normal anatomical landmarks become obscured by the diffuse infiltration. Imaging studies reveal characteristic features including loss of the normal fibrillar pattern, increased tendon thickness with maintained echogenicity, and the absence of increased vascularity that would suggest inflammatory tendinopathy. Patients with hereditary amyloidosis, particularly those with transthyretin mutations, may show bilateral Achilles tendon involvement as an early manifestation of systemic disease, making recognition of these changes crucial for early diagnosis and treatment. The metabolic implications of amyloidosis extend beyond the local tendon effects, as patients often develop cardiac involvement, renal dysfunction, and neuropathy that require comprehensive management and monitoring.