8 Eye Appearance Changes Associated with Systemic Conditions
The human eye serves as far more than a mere organ of vision; it functions as a remarkable diagnostic window that can reveal the presence of numerous systemic conditions affecting the entire body. Throughout medical history, astute physicians have recognized that ocular manifestations often precede or accompany systemic diseases, making ophthalmologic examination an invaluable diagnostic tool. The transparent nature of ocular tissues, combined with the eye's rich vascular supply and neural connections, creates a unique opportunity for healthcare providers to observe pathological changes that mirror processes occurring elsewhere in the body. From subtle changes in pupil reactivity to dramatic alterations in retinal appearance, the eye's various structures can exhibit telltale signs of diabetes, hypertension, autoimmune disorders, neurological conditions, and metabolic diseases. This comprehensive exploration will examine eight distinct eye appearance changes that serve as important indicators of systemic health conditions, demonstrating how careful observation of ocular signs can lead to early detection, proper diagnosis, and timely intervention for potentially serious medical conditions that extend far beyond the eye itself.
1. Diabetic Retinopathy - The Sweet Destruction

Diabetic retinopathy represents one of the most significant and well-documented examples of how systemic disease manifests in ocular appearance, serving as both a complication and an early warning sign of poorly controlled diabetes mellitus. This progressive condition develops when chronically elevated blood glucose levels damage the delicate retinal blood vessels, leading to a cascade of pathological changes that are readily visible during ophthalmoscopic examination. In the early stages, known as non-proliferative diabetic retinopathy, healthcare providers can observe microaneurysms appearing as small red dots, flame-shaped hemorrhages, hard exudates that appear as yellow waxy deposits, and cotton wool spots indicating nerve fiber layer infarcts. As the condition progresses to proliferative diabetic retinopathy, new abnormal blood vessels begin growing on the retinal surface and into the vitreous cavity, creating a characteristic appearance of neovascularization that can be accompanied by fibrous tissue formation. The presence and severity of diabetic retinopathy correlate strongly with the duration and control of diabetes, making regular eye examinations crucial for diabetic patients. Advanced cases may show vitreous hemorrhage, retinal detachment, and macular edema, all of which can severely impact vision while simultaneously indicating the need for aggressive systemic diabetes management to prevent further complications.
2. Hypertensive Retinopathy - Pressure's Visual Impact

Hypertensive retinopathy demonstrates how chronic high blood pressure leaves its unmistakable signature on the retinal vasculature, creating a spectrum of appearance changes that directly correlate with the severity and duration of systemic hypertension. The retinal arterioles, being among the smallest vessels in the body that can be directly visualized, serve as excellent indicators of systemic vascular health and the effects of elevated blood pressure throughout the circulatory system. In mild hypertensive retinopathy, ophthalmoscopic examination reveals arterial narrowing, increased arterial light reflex giving vessels a "copper wire" or "silver wire" appearance, and arteriovenous nicking where arteries compress underlying veins at crossing points. As hypertension becomes more severe and chronic, the retinal changes become increasingly dramatic, progressing to include flame-shaped hemorrhages, cotton wool spots, hard exudates, and in malignant hypertension, papilledema with optic disc swelling. The Keith-Wagener-Barker classification system grades these changes from mild (Grade I) to severe (Grade IV), with higher grades indicating more severe systemic hypertension and increased cardiovascular risk. These retinal findings often appear before patients experience symptoms of hypertensive complications elsewhere in the body, making ophthalmoscopic examination a valuable tool for assessing cardiovascular risk and monitoring the effectiveness of antihypertensive treatment regimens.
3. Thyroid Eye Disease - When Hormones Affect Vision

Thyroid eye disease, also known as Graves' ophthalmopathy, creates distinctive and often dramatic changes in eye appearance that reflect the complex autoimmune processes associated with thyroid dysfunction, particularly hyperthyroidism caused by Graves' disease. This condition results from the cross-reactivity of thyroid-stimulating immunoglobulins with tissues in the orbit, leading to inflammation, fibrosis, and enlargement of the extraocular muscles and orbital fat. The most characteristic appearance changes include upper and lower eyelid retraction, creating a "staring" appearance with excessive white sclera visible above and below the iris, proptosis or forward protrusion of the eyes due to increased orbital tissue volume, and periorbital edema that gives the eyes a puffy, swollen appearance. Patients may also develop conjunctival injection and chemosis, where the conjunctiva appears red and swollen, sometimes to the point of protruding beyond the eyelid margins. The extraocular muscle involvement can lead to diplopia and restricted eye movements, while severe cases may progress to compressive optic neuropathy, threatening vision. The severity of thyroid eye disease doesn't always correlate directly with thyroid hormone levels, and the condition can sometimes precede, accompany, or follow the diagnosis of thyroid dysfunction. Recognition of these ocular signs is crucial because early intervention with immunosuppressive therapy can help prevent permanent structural changes and preserve both function and appearance.
4. Wilson's Disease - The Copper Ring Mystery

Wilson's disease, a rare genetic disorder affecting copper metabolism, produces one of the most pathognomonic ocular signs in medicine through the formation of Kayser-Fleischer rings, distinctive copper deposits that create a characteristic golden-brown or greenish ring around the corneal periphery. These rings result from the accumulation of copper in Descemet's membrane of the cornea due to the body's inability to properly excrete excess copper, leading to toxic accumulation in various organs including the liver, brain, and eyes. The Kayser-Fleischer rings typically begin as deposits in the superior and inferior aspects of the corneal periphery before progressing to form complete rings, and they are best visualized using slit-lamp examination, though advanced cases may be visible to the naked eye. While these rings are present in virtually all patients with neurological manifestations of Wilson's disease, they can also be found in approximately 50-65% of patients with purely hepatic presentations, making them an invaluable diagnostic marker for this treatable condition. The intensity and completeness of the rings generally correlate with the severity of copper accumulation and may diminish with effective chelation therapy using medications like penicillamine or trientine. Beyond the Kayser-Fleischer rings, patients with Wilson's disease may also develop sunflower cataracts, which appear as multicolored, petal-like deposits in the lens, though these are less common and typically occur in more advanced cases. Early recognition of these ocular signs is crucial because Wilson's disease is progressive and potentially fatal if left untreated, but responds well to copper chelation therapy when diagnosed early.
5. Autoimmune Conditions - When the Body Attacks the Eyes
Autoimmune conditions create a diverse array of ocular manifestations that reflect the complex interplay between systemic inflammation and ocular tissues, with appearance changes ranging from subtle inflammatory signs to dramatic structural alterations. Rheumatoid arthritis, one of the most common autoimmune conditions, can cause keratoconjunctivitis sicca (dry eye syndrome), leading to a dull, lackluster appearance of the ocular surface, conjunctival injection, and in severe cases, corneal thinning or perforation that creates irregular corneal contours. Systemic lupus erythematosus presents with cotton wool spots, retinal hemorrhages, and in cases of associated antiphospholipid syndrome, retinal vascular occlusions that can dramatically alter retinal appearance. Sjögren's syndrome, an autoimmune condition targeting moisture-producing glands, creates characteristic dry eye changes with reduced tear film, conjunctival staining patterns visible with fluorescein dye, and a generally irritated, red appearance of the ocular surface. Behçet's disease can cause recurrent uveitis with hypopyon, creating a distinctive layering of white inflammatory cells in the anterior chamber that is visible as a white or yellow fluid level at the bottom of the iris. Vogt-Koyanagi-Harada disease, an autoimmune condition affecting melanocytes, can cause bilateral granulomatous uveitis with characteristic "mutton-fat" keratic precipitates on the corneal endothelium, giving the cornea a distinctive spotted appearance. These autoimmune-related ocular changes often fluctuate with disease activity and may be among the earliest manifestations of systemic autoimmune conditions, making regular ophthalmologic monitoring essential for patients with known autoimmune diseases and valuable for detecting these conditions in their early stages.
6. Neurological Disorders - The Brain-Eye Connection
Neurological disorders create distinctive patterns of ocular appearance changes that reflect the intimate connection between the brain and visual system, with pupillary abnormalities, eyelid positioning changes, and eye movement disorders serving as important diagnostic clues for various neurological conditions. Horner's syndrome, resulting from sympathetic nerve pathway disruption, creates a classic triad of ptosis (drooping eyelid), miosis (constricted pupil), and anhidrosis (decreased sweating), giving the affected eye a noticeably smaller and more closed appearance compared to the unaffected side. Multiple sclerosis can cause internuclear ophthalmoplegia, leading to characteristic eye movement abnormalities where one eye fails to adduct properly during horizontal gaze, creating an asymmetric appearance during eye movements and often accompanied by nystagmus in the abducting eye. Myasthenia gravis, an autoimmune neuromuscular disorder, typically presents with variable ptosis that worsens with fatigue, creating a droopy, tired appearance that may fluctuate throughout the day and can affect one or both eyes asymmetrically. Third cranial nerve palsy, whether from diabetes, aneurysm, or other causes, creates a dramatic appearance with complete ptosis, a dilated pupil, and the eye positioned "down and out" due to unopposed action of the fourth and sixth cranial nerves. Parkinson's disease can cause reduced blink rate and decreased facial expression, giving the eyes a fixed, staring appearance, while also potentially causing convergence insufficiency and difficulty with voluntary eye movements. Progressive supranuclear palsy creates characteristic vertical gaze limitations, particularly affecting downward eye movements, which becomes apparent during clinical examination and can significantly alter the patient's ability to maintain normal eye contact and visual interaction patterns.
7. Liver Disease - Hepatic Reflections in the Eyes

Liver disease creates several distinctive ocular appearance changes that reflect the liver's crucial role in metabolism, detoxification, and protein synthesis, with these changes often serving as important indicators of hepatic dysfunction severity. Jaundice, perhaps the most recognizable sign of liver disease, causes yellowing of the sclera (icterus) due to elevated bilirubin levels, with the scleral icterus often being more apparent than skin yellowing and typically becoming visible when serum bilirubin levels exceed 2-3 mg/dL. Wilson's disease, as previously discussed, creates pathognomonic Kayser-Fleischer rings, but other forms of liver disease can also affect ocular appearance through different mechanisms. Chronic liver disease and cirrhosis can lead to decreased albumin production, resulting in periorbital edema that gives the eyes a puffy, swollen appearance, while portal hypertension may cause conjunctival vessel dilation and a generally more vascular appearance of the ocular surface. Primary biliary cirrhosis, an autoimmune liver condition, is frequently associated with dry eye syndrome and can cause xanthelasma, yellowish cholesterol deposits around the eyelids that create distinctive plaques or nodules in the periorbital area. Acute hepatitis can cause photophobia and conjunctival injection, giving the eyes a red, irritated appearance that may be accompanied by general malaise and systemic symptoms. Hepatic encephalopathy, a complication of advanced liver disease, can cause altered mental status that may be reflected in changes to pupillary reactivity, eye movement coordination, and overall ocular responsiveness. The presence and severity of these ocular changes often correlate with the degree of hepatic dysfunction and can provide valuable information about disease progression and the need for more aggressive medical intervention or consideration of liver transplantation.
8. Cardiovascular Disease - Vascular Changes Visible in the Eye
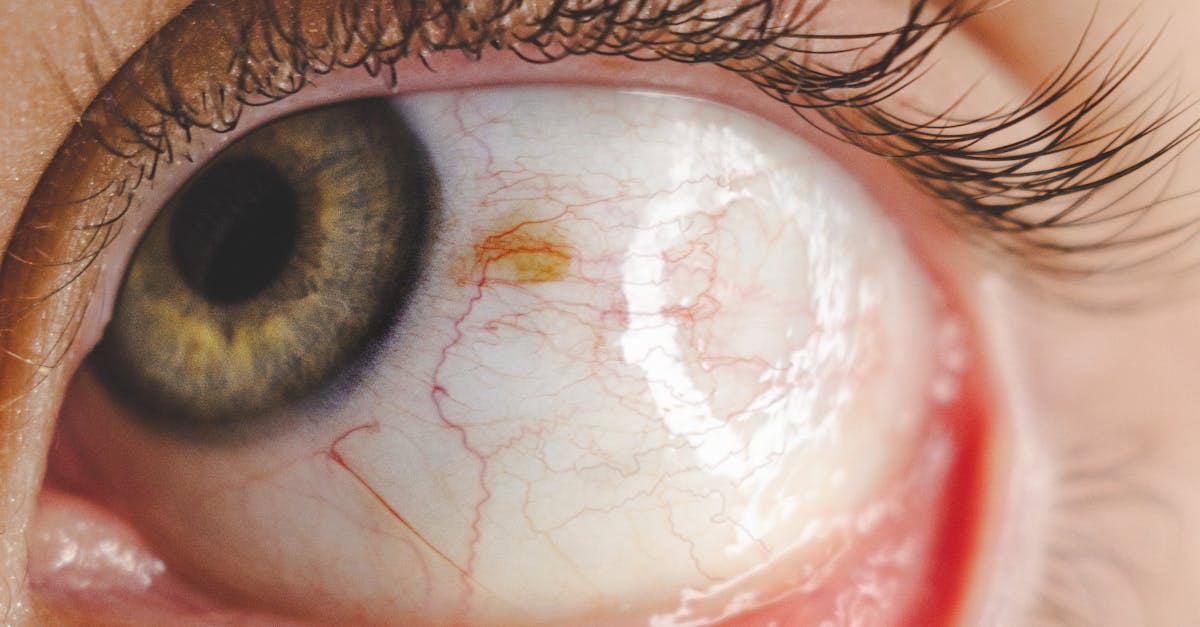
Cardiovascular disease manifests in the eye through various vascular changes that provide a unique window into the health of the systemic circulatory system, with retinal vessel appearance serving as an accessible indicator of cardiovascular risk and disease progression. Atherosclerotic changes in the retinal arterioles mirror those occurring in coronary and cerebral vessels, with arterial narrowing, increased tortuosity, and changes in the arterial light reflex providing early indicators of systemic vascular disease. Embolic phenomena from carotid artery disease or cardiac sources can cause retinal artery occlusions, leading to sudden, dramatic changes in retinal appearance with areas of retinal whitening, cherry-red spots at the macula, and visible emboli within retinal vessels that appear as bright, refractile plugs. Hollenhorst plaques, cholesterol emboli that appear as bright, yellow-orange refractile spots at arterial bifurcations, serve as important markers of carotid artery disease and increased stroke risk, often being discovered during routine eye examinations before patients experience neurological symptoms. Central retinal vein occlusion, often associated with cardiovascular risk factors, creates a dramatic "blood and thunder" fundus appearance with extensive retinal hemorrhages, cotton wool spots, and optic disc swelling that reflects the severity of retinal vascular compromise. The caliber and appearance of retinal vessels have been shown to correlate with cardiovascular mortality, with narrower arterioles and wider venules associated with increased risk of coronary heart disease and stroke. Advanced cardiovascular disease may also manifest as ischemic optic neuropathy, causing optic disc pallor and visual field defects that reflect compromised blood supply to the optic nerve, while systemic hypotension can cause watershed infarcts in the retina, creating characteristic patterns of retinal ischemia that mirror similar processes in the brain and other organs.
9. Integrating Ocular Signs into Comprehensive Healthcare

The eight eye appearance changes associated with systemic conditions discussed throughout this exploration underscore the critical importance of incorporating comprehensive ocular examination into routine healthcare assessment and the management of systemic diseases. From the progressive vascular changes of diabetic and hypertensive retinopathy to the distinctive copper deposits of Wilson's disease, these ocular manifestations serve as invaluable diagnostic tools that can facilitate early detection, monitor disease progression, and guide therapeutic interventions for conditions that extend far beyond the eye itself. The accessibility of ocular examination, combined with the transparency of ocular tissues and the eye's rich vascular and neural connections, creates unique opportunities for healthcare providers to observe pathological processes that would otherwise require invasive procedures or advanced imaging techniques to detect. Modern healthcare's increasing emphasis on preventive medicine and early intervention makes the recognition of these ocular signs even more crucial, as many of the systemic conditions discussed can be effectively managed or even reversed when detected in their early stages. Furthermore, the correlation between ocular findings and systemic disease severity provides valuable prognostic information that can help guide treatment intensity and monitoring frequency. As medical technology continues to advance, new imaging techniques and diagnostic tools are enhancing our ability to detect subtle ocular changes even earlier, potentially revolutionizing our approach to screening and managing systemic diseases. The integration of artificial intelligence and machine learning into ophthalmologic practice promises to further expand our diagnostic capabilities, making the eye an even more powerful window into systemic health. Ultimately, the recognition that ocular appearance changes can reflect systemic conditions emphasizes the interconnected nature of human physiology and the importance of comprehensive, multidisciplinary healthcare approaches that consider the eye not as an isolated organ, but as an integral component of overall health assessment and disease management.